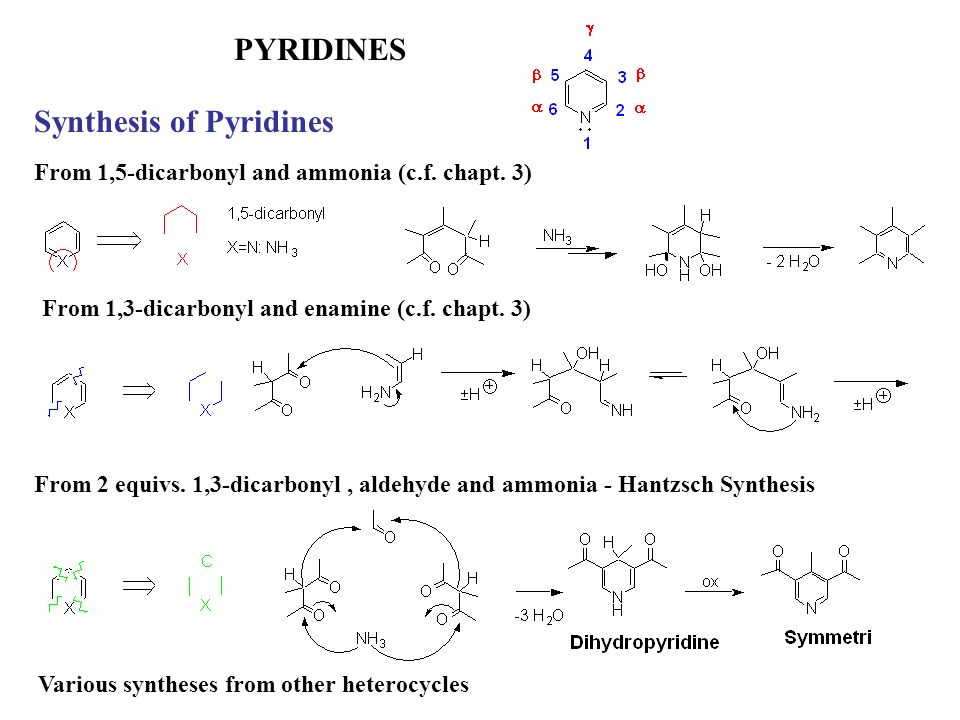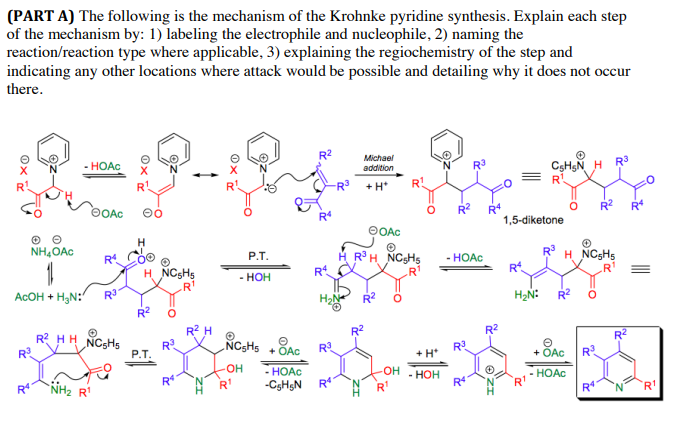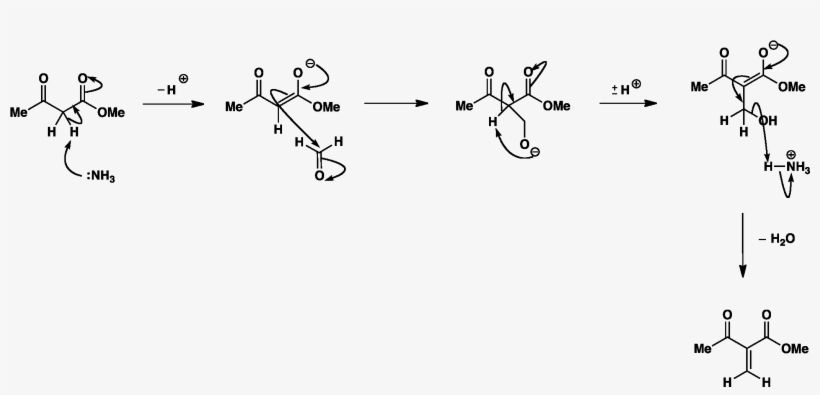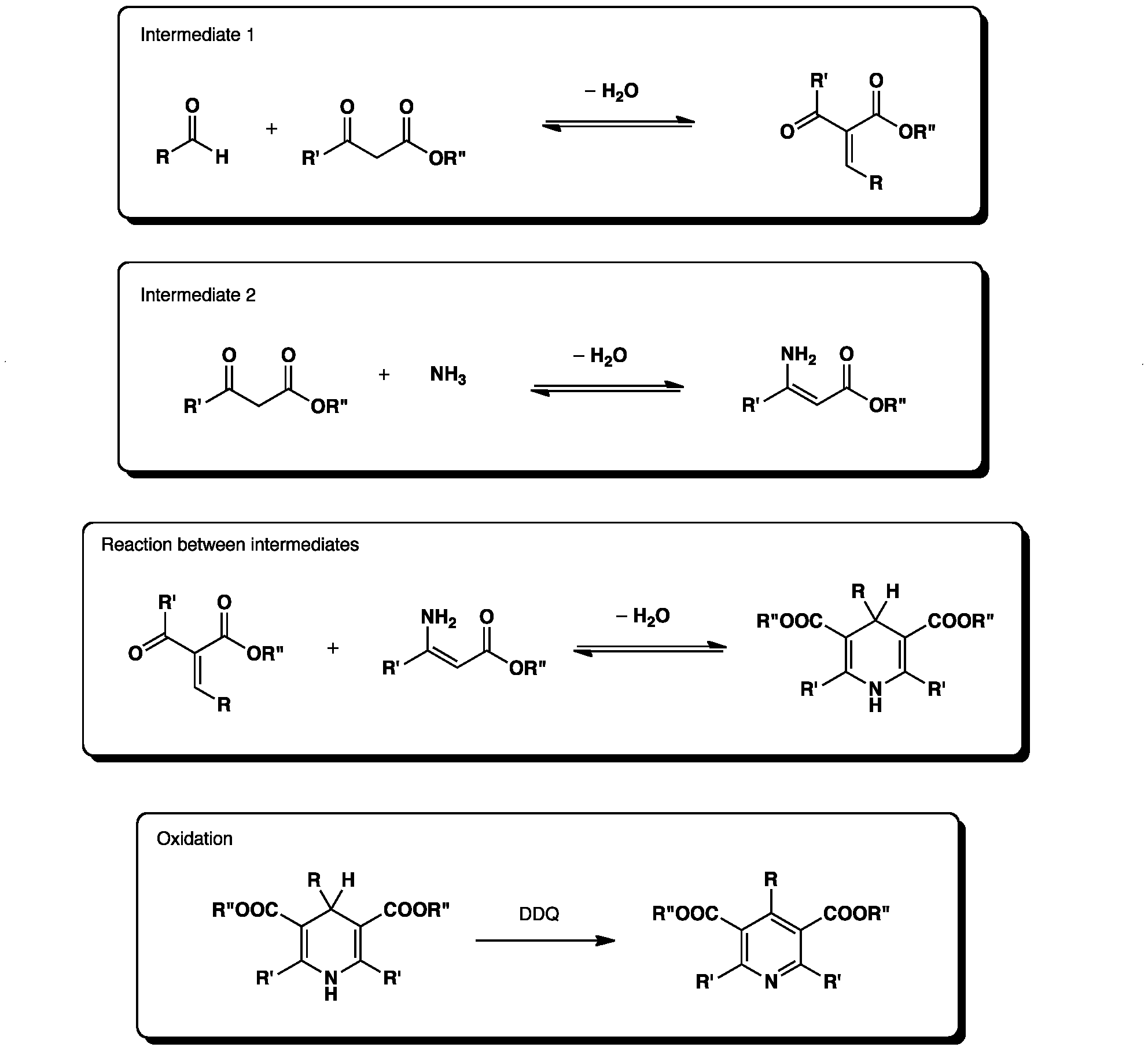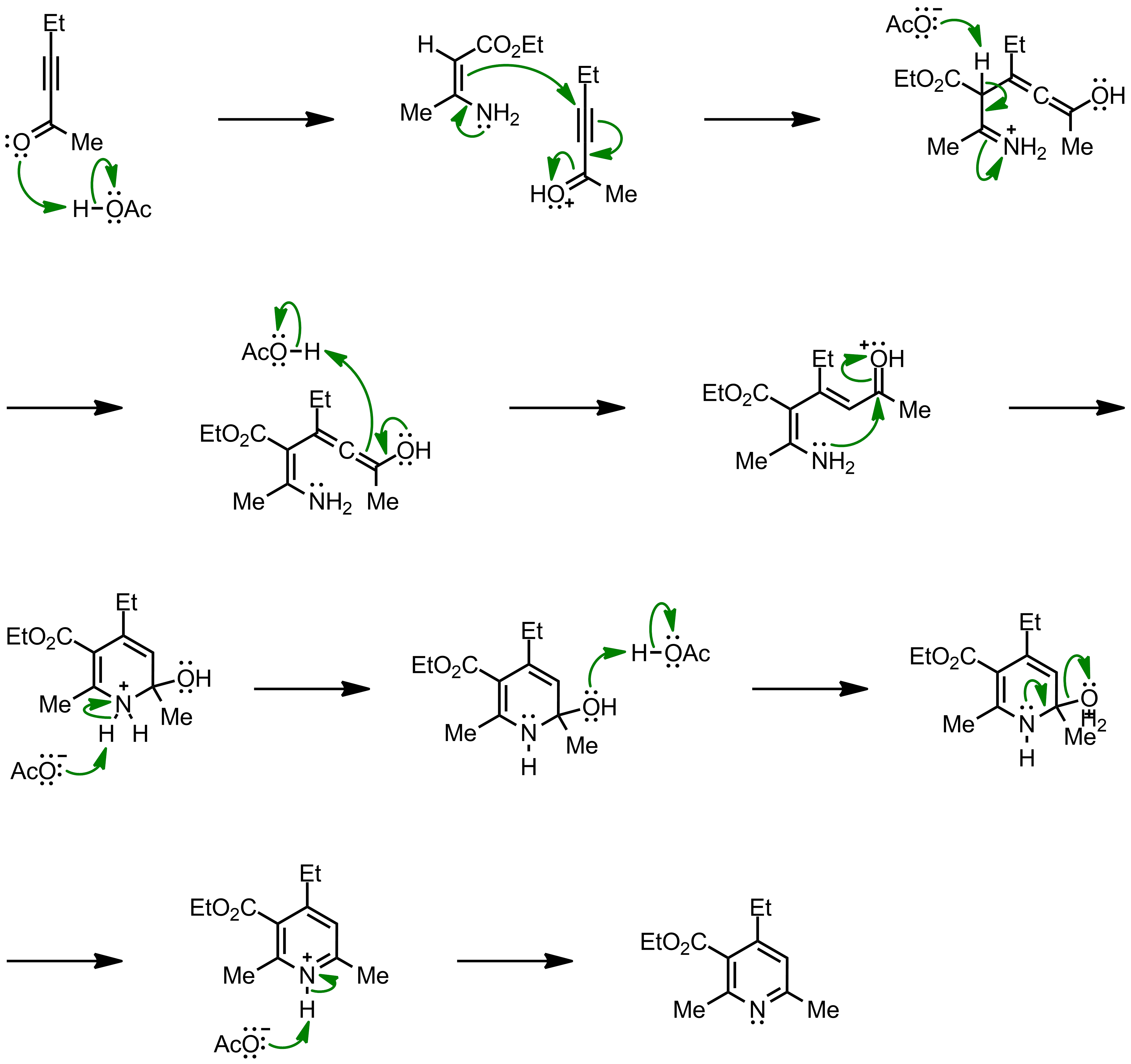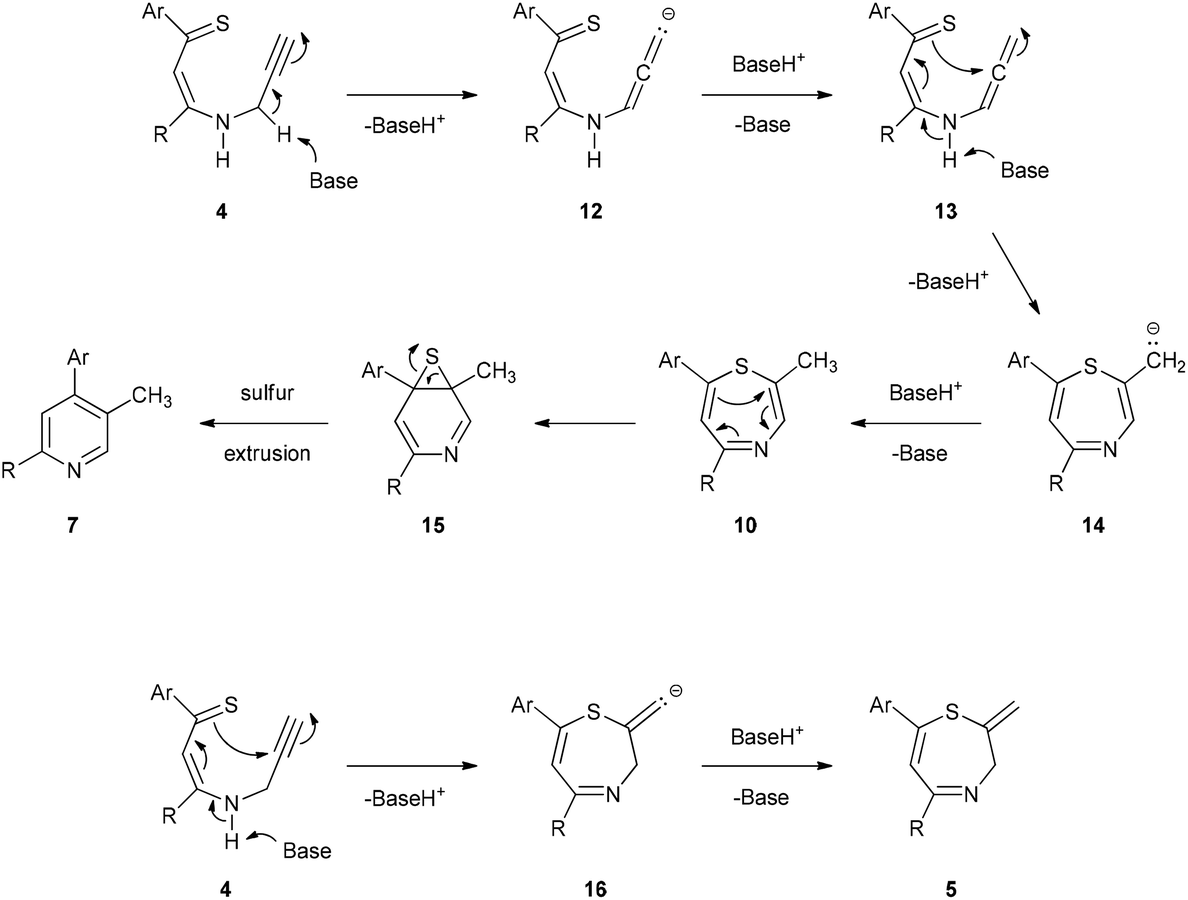
A new strategy for the synthesis of pyridines from N -propargylic β-enaminothiones - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB03180K
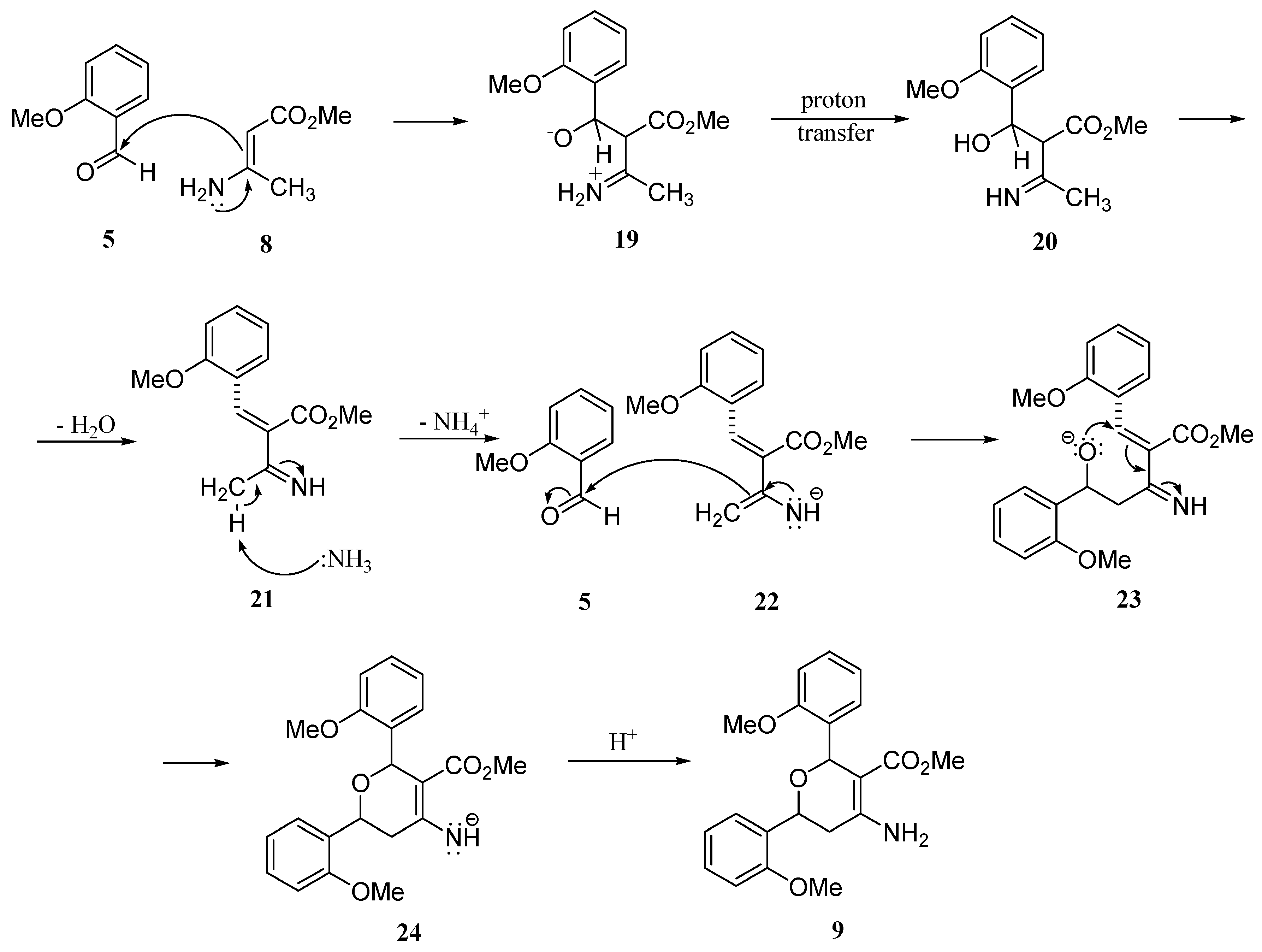
Molecules | Free Full-Text | Hantzsch Synthesis of 2,6-Dimethyl-3,5-dimethoxycarbonyl-4-(o-methoxyphenyl)-1,4-dihydropyridine; a Novel Cyclisation Leading to an Unusual Formation of 1-Amino-2-methoxycarbonyl-3,5-bis(o-methoxyphenyl)-4-oxa-cyclohexan-1 ...
Synthesis, Reactions and Medicinal Uses of Pyridine and Basicity of Pyridine : Pharmaceutical Guidelines

Synthesis and antibacterial activity of novel pyridine and pyrazine derivatives obtained from amidoximes - Gobis - 2009 - Journal of Heterocyclic Chemistry - Wiley Online Library
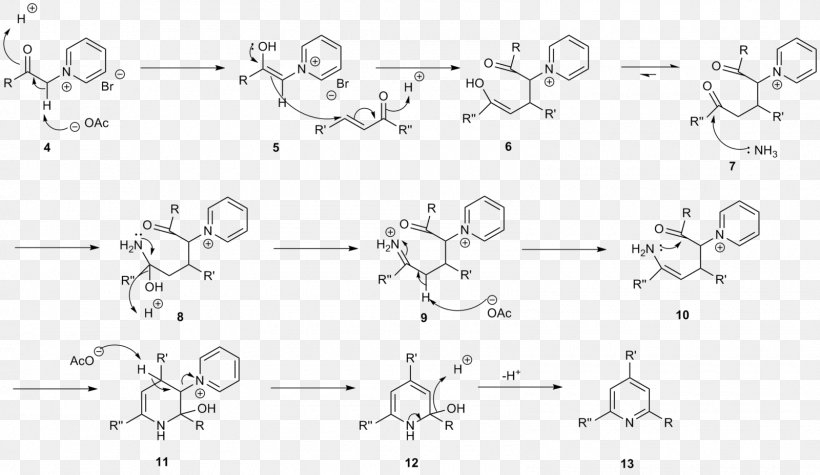
Kröhnke Pyridine Synthesis Hantzsch Pyridine Synthesis Chemical Synthesis 2,6-Lutidine, PNG, 1600x928px, Watercolor, Cartoon, Flower,

Kröhnke pyridine synthesis Hantzsch pyridine synthesis Chemical synthesis 2,6-Lutidine, salt, angle, white, text png | PNGWing
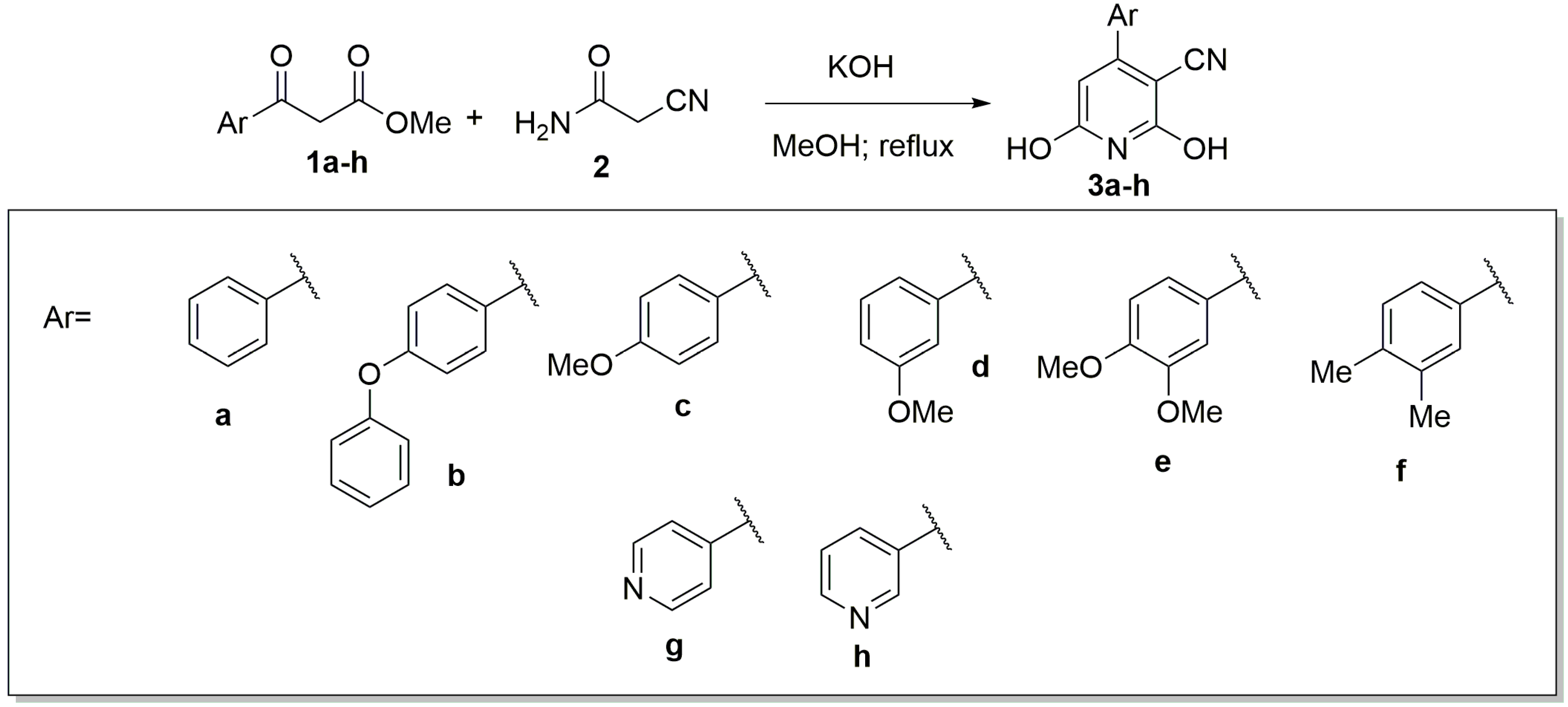
Molecules | Free Full-Text | The Guareschi Pyridine Scaffold as a Valuable Platform for the Identification of Selective PI3K Inhibitors | HTML
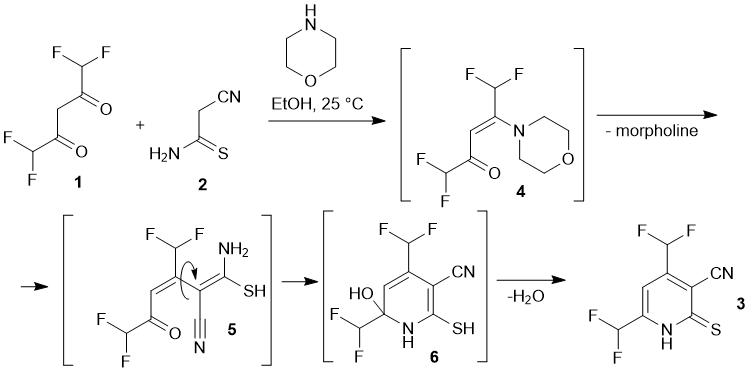

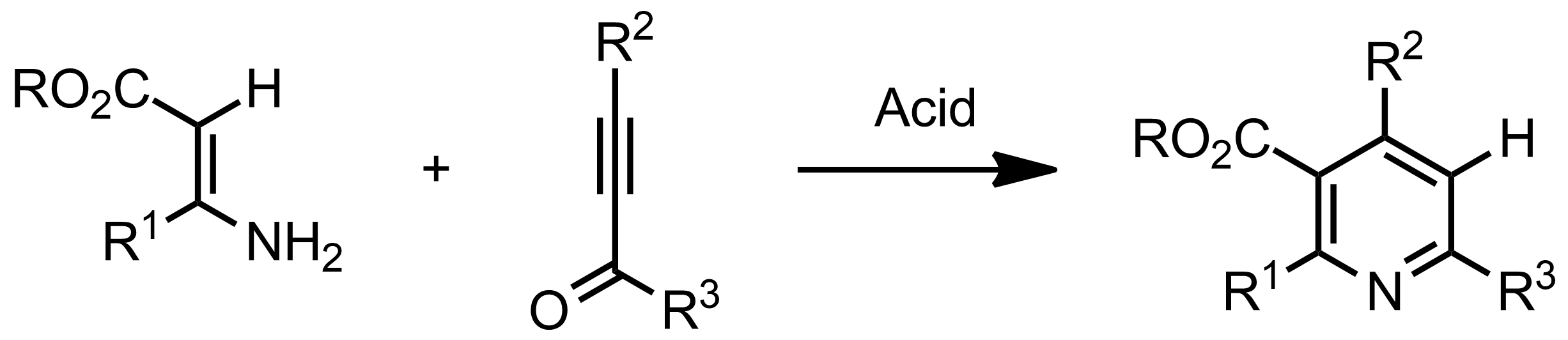
.gif)