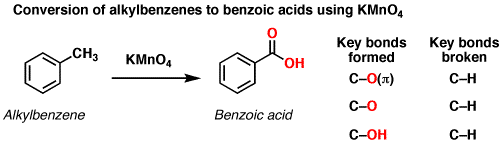
Reaction For Removal of Vinyl Chloride Using Potassium Permanganate | HS-600 | Hydrosil International

Kinetics and mechanisms of oxidation of d-fructose and d-lactose by permanganate ion in acidic medium
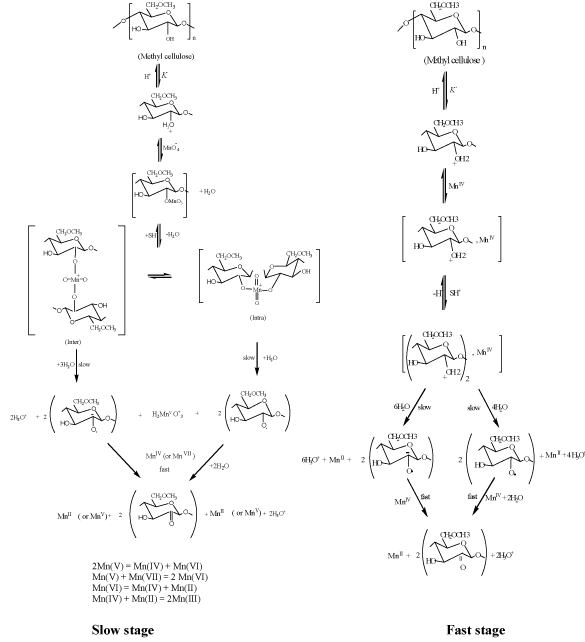
Novel synthesis of coordination biopolymer precursor from oxidation of methyl cellulose by alkaline potassium permanganate

Chem 52 (W03): Demonstration of Alcohol Oxidation Three different types of alcohols (1º, 2º, 3º) are oxidized with basic potassium permanganate (MnO4- /HO-). The amount of MnO2 formed gives a colorimetric indication of the extent to which each alcohol is ...
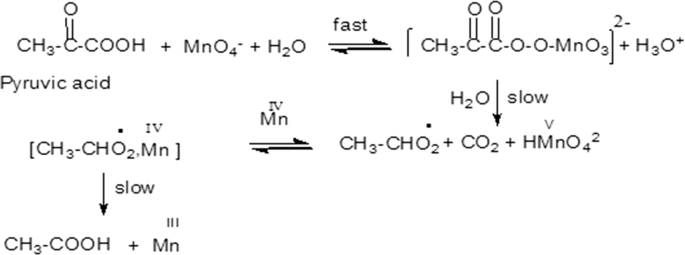
Kinetics and mechanism of oxidation of pyruvate by permanganate ion in aqueous perchlorate solution | SpringerLink
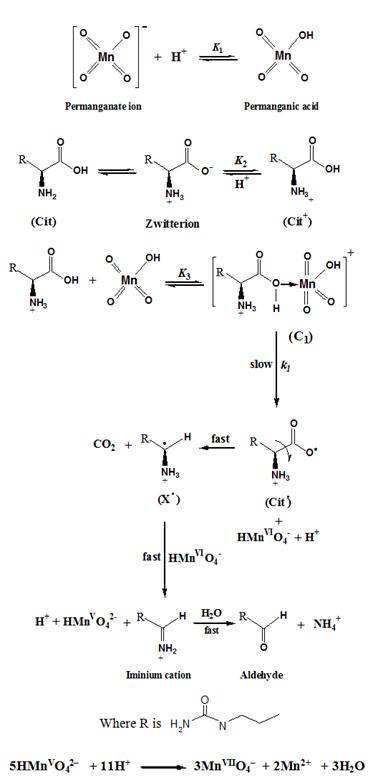
Kinetics and Mechanistic Study of Permanganate Oxidation of L-Citrulline in Acidic and Basic Media :: Science Publishing Group

Kinetics and Mechanistic Study of Permanganate Oxidation of L-Citrulline in Acidic and Basic Media :: Science Publishing Group




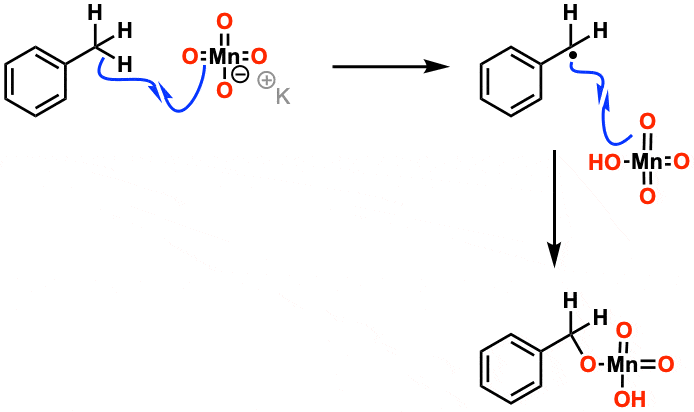
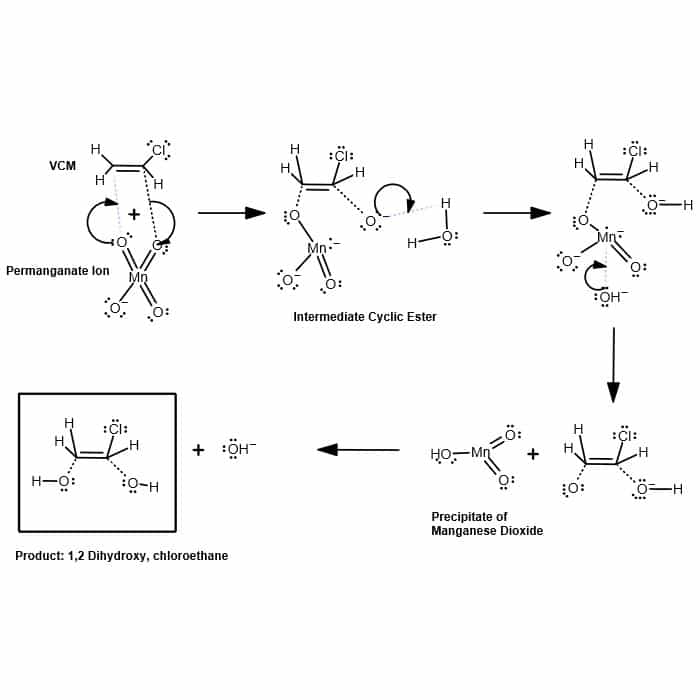
/chapter9/pages21and22/page21and22_files/diol_KMno4_mech.png)
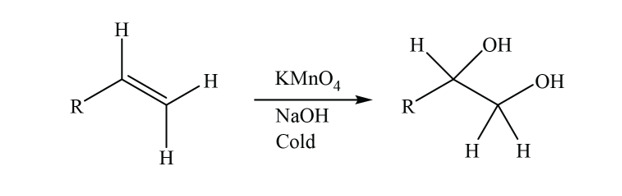


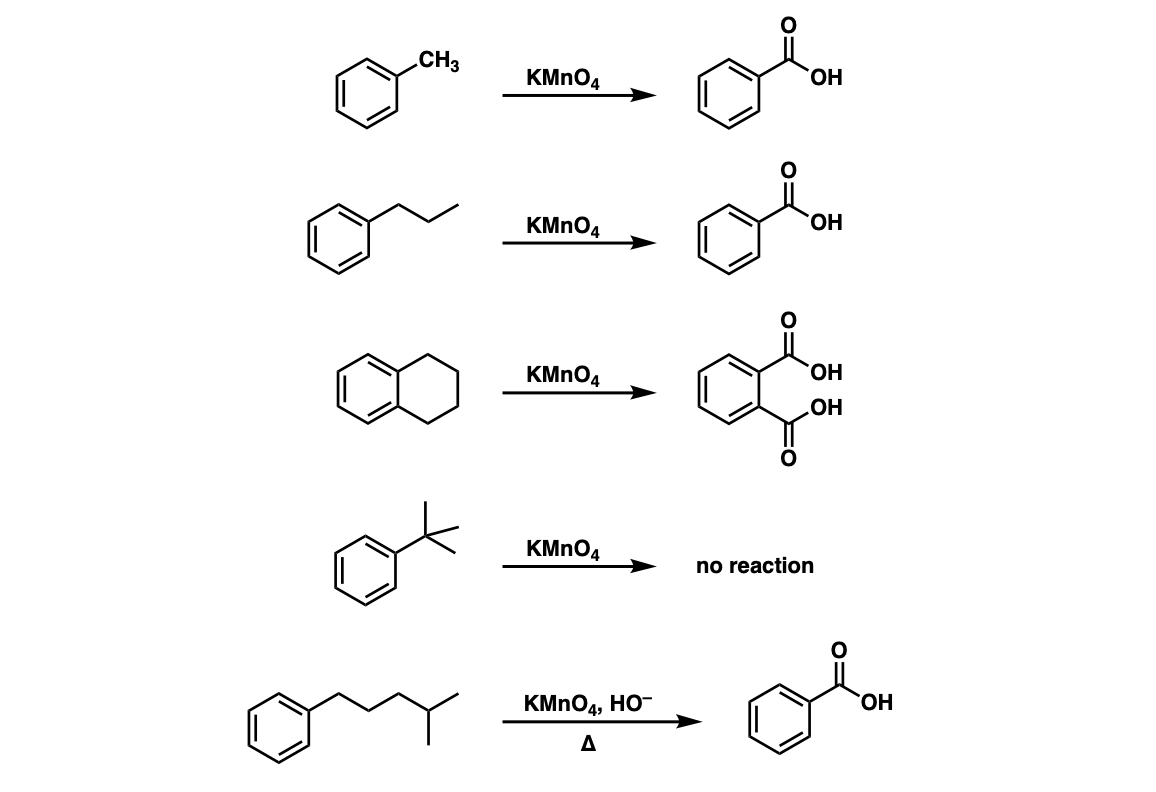


![Oxidation to carboxylic acid [H2CrO4 or KMnO4] - ChemistryScore Oxidation to carboxylic acid [H2CrO4 or KMnO4] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Oxidation-to-carboxylic-acid-H2CrO4-or-KMnO43-768x431.png)
![PDF] The mechanism of permanganate oxidation of sulfides and sulfoxides. | Semantic Scholar PDF] The mechanism of permanganate oxidation of sulfides and sulfoxides. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/18e51e648e71181d0f214f4b4876dce29f61d944/1-Figure1-1.png)
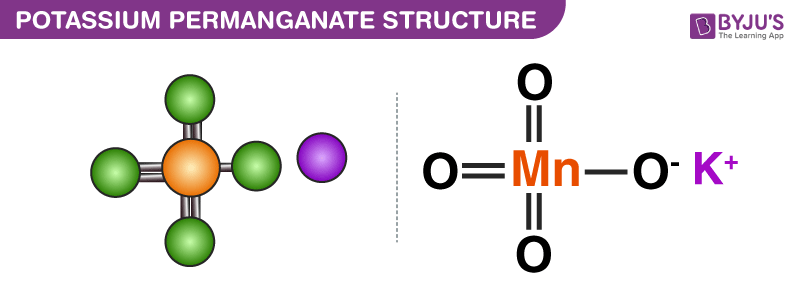
![PDF] Oxidation by permanganate: synthetic and mechanistic aspects | Semantic Scholar PDF] Oxidation by permanganate: synthetic and mechanistic aspects | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fe91f5161ca1ab481ae111f1abc82d37d336f021/15-Figure2-1.png)
![Oxidative Cleavage [KMnO4] - ChemistryScore Oxidative Cleavage [KMnO4] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Oxidative-cleavage1-768x350.png)