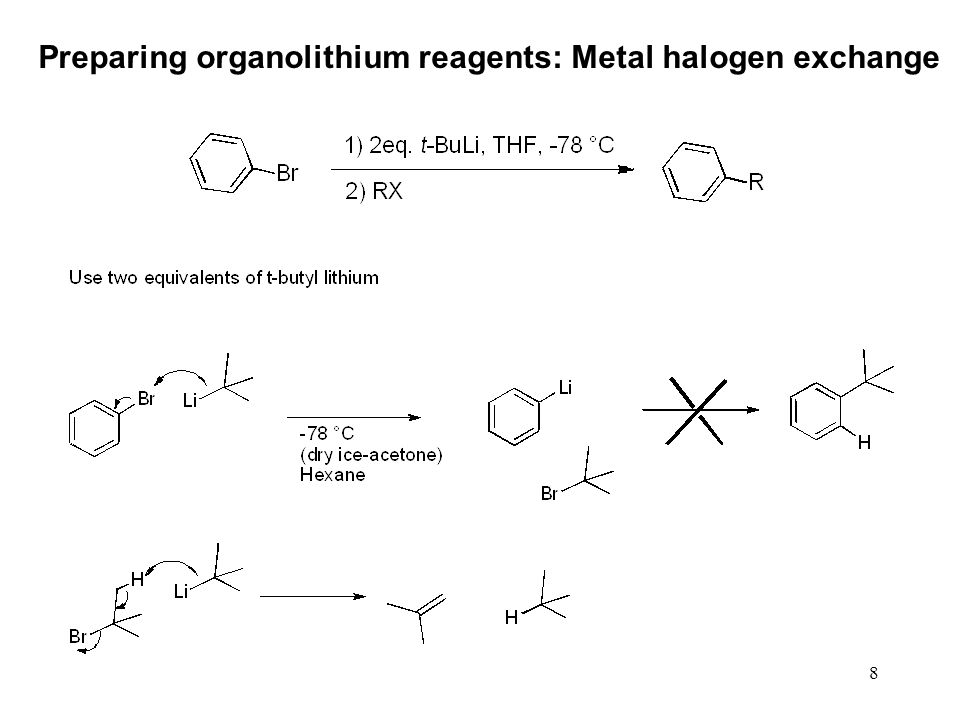
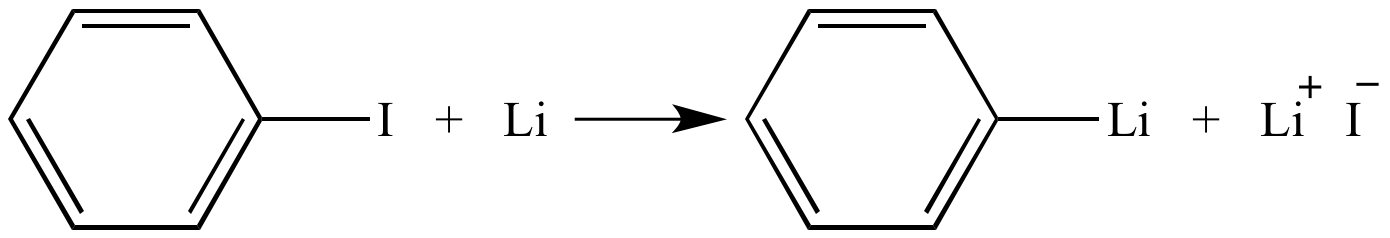
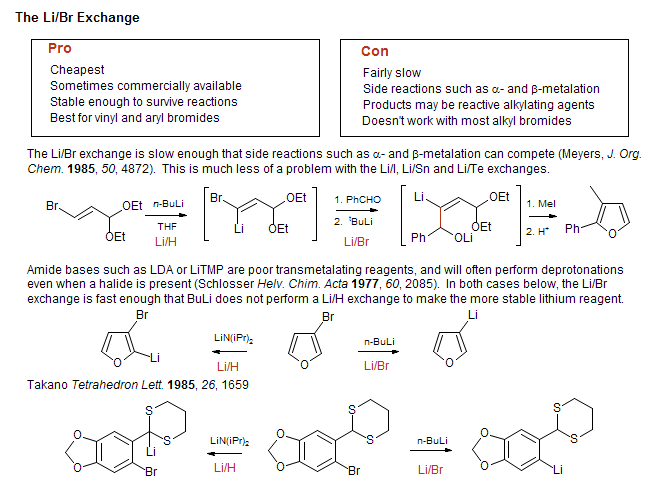
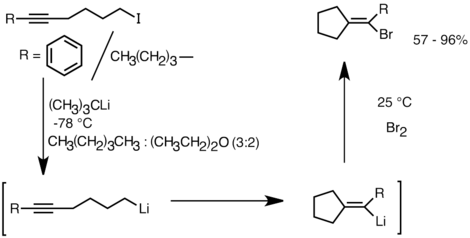
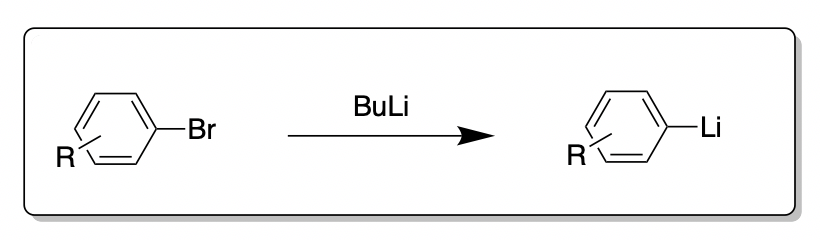
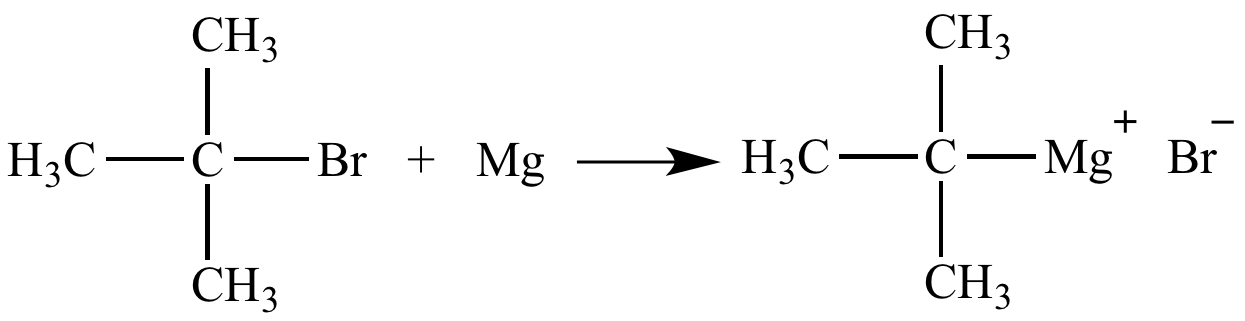Lithium–Bromide Exchange versus Nucleophilic Addition of Schiff's base: Unprecedented Tandem Cyclisation Pathways - Orr - 2019 - Chemistry – A European Journal - Wiley Online Library
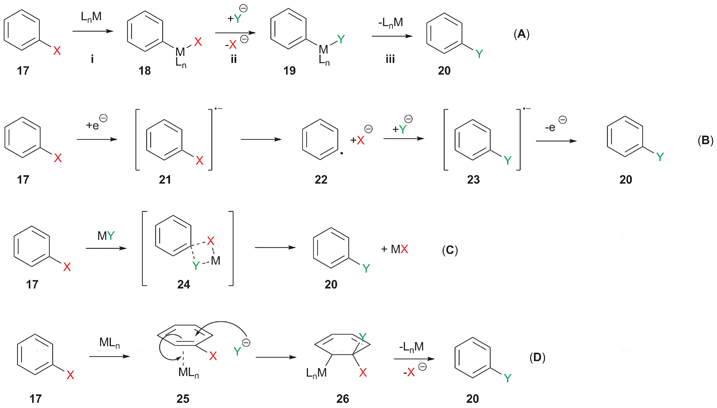
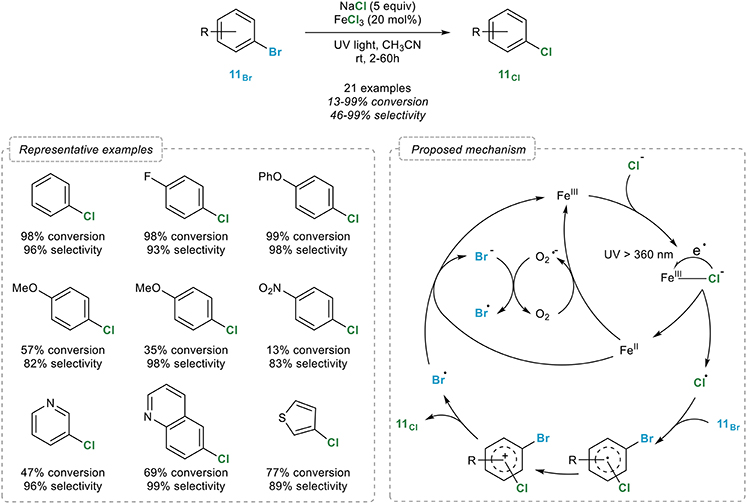
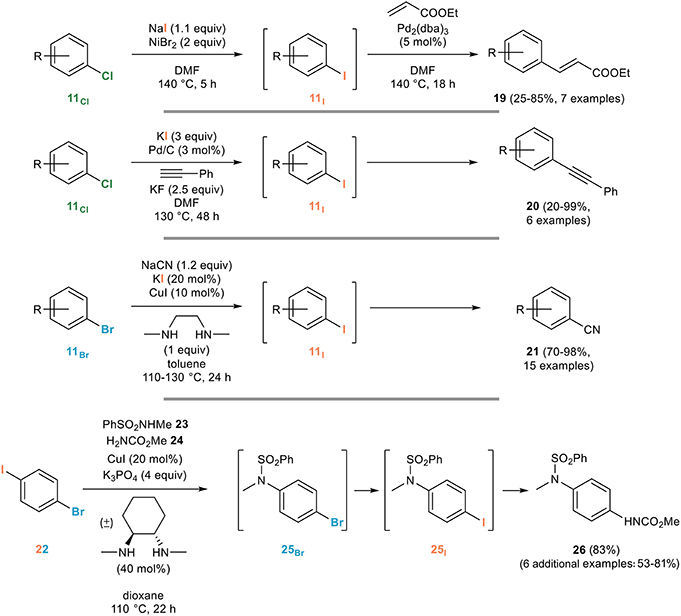
Metal-catalysed halogen exchange reactions of aryl halides - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B818155A

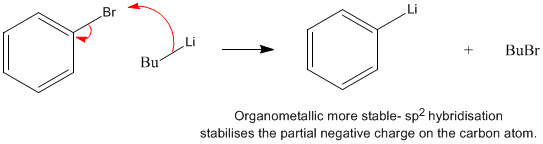
Selective Halogen−Magnesium Exchange Reaction via Organomagnesium Ate Complex | The Journal of Organic Chemistry

Recent Advances of the Halogen–Zinc Exchange Reaction - Balkenhohl - 2020 - Chemistry – A European Journal - Wiley Online Library

Halogen–lithium exchange versus deprotonation: synthesis of diboronic acids derived from aryl–benzyl ethers - ScienceDirect
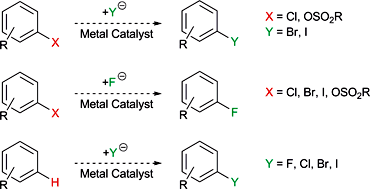
Metal-catalysed halogen exchange reactions of aryl halides - Organic & Biomolecular Chemistry (RSC Publishing)

Halogen exchange in alkyl halides: the Swarts and Finkelstein reactions. | Download Scientific Diagram
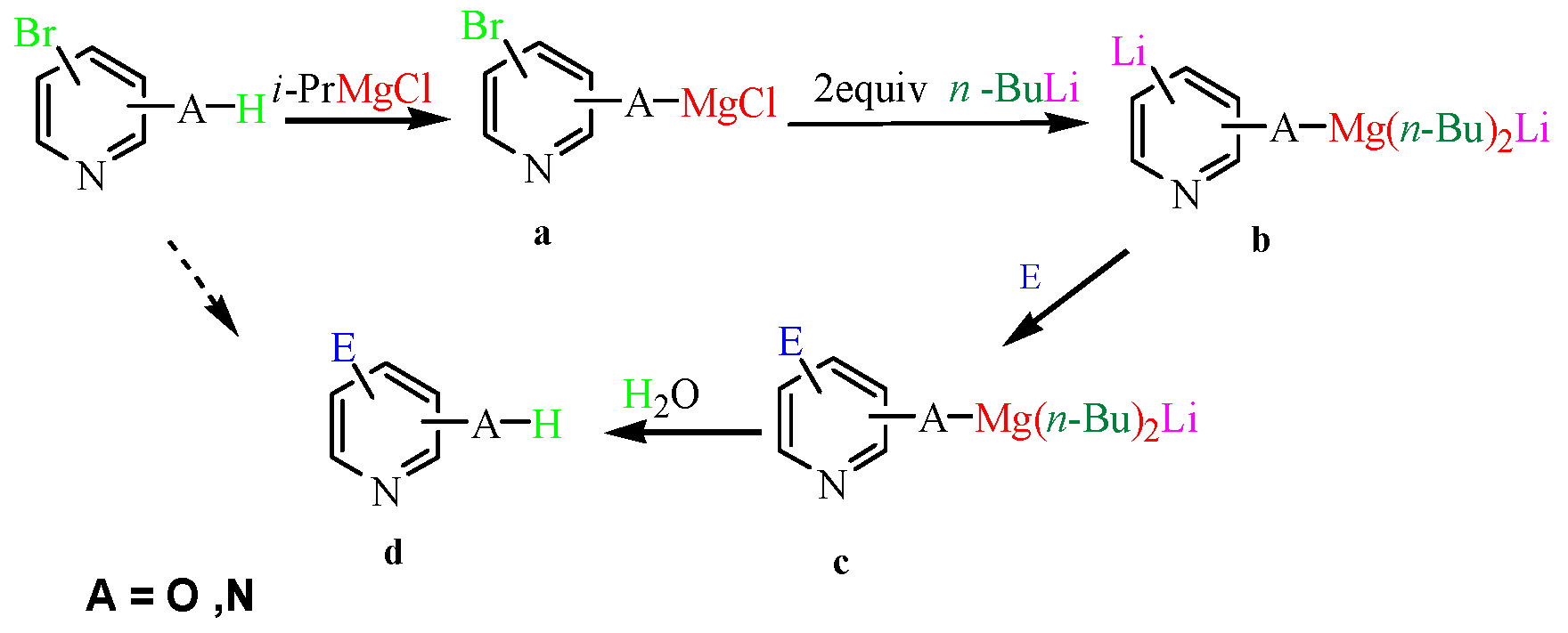
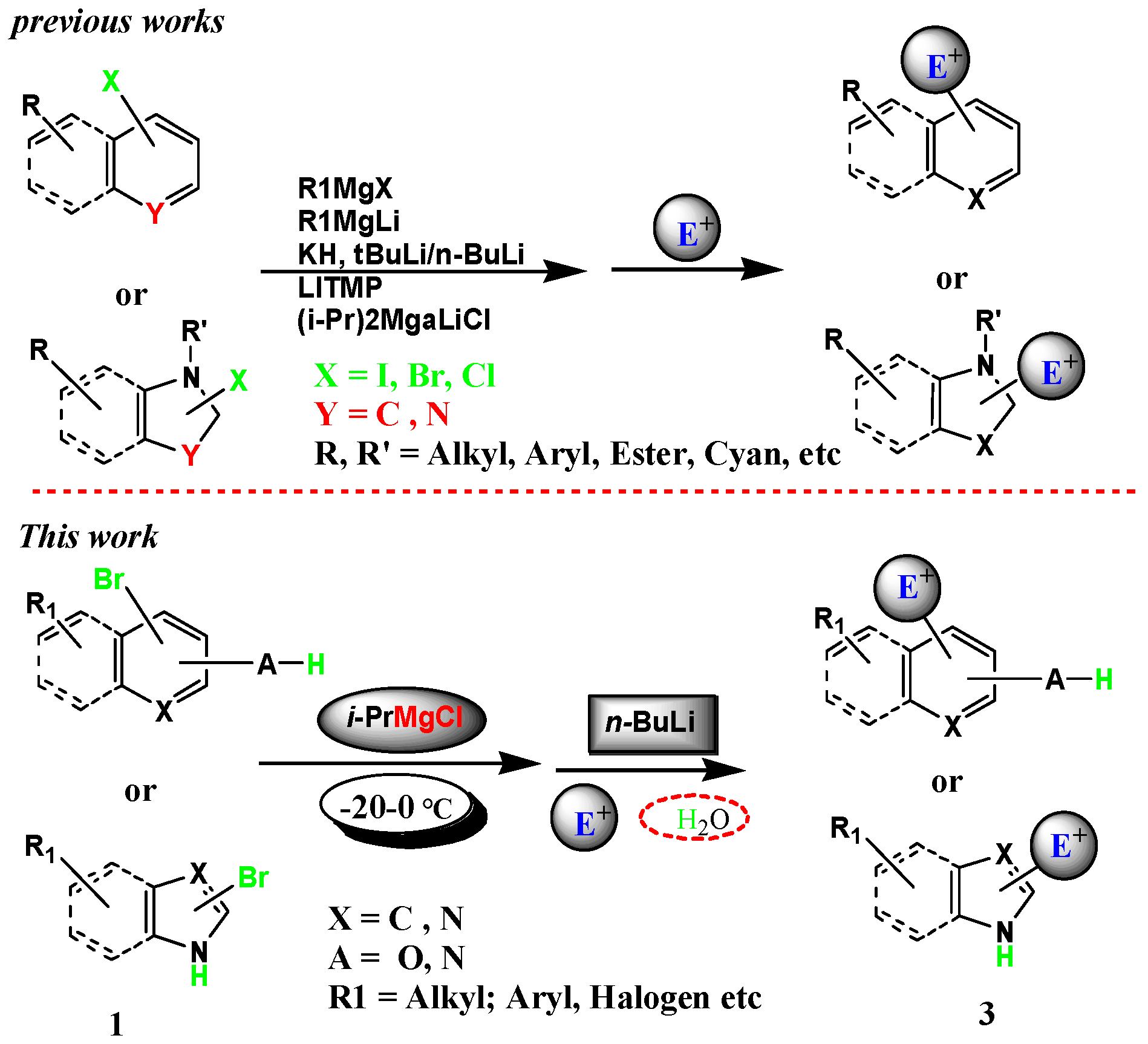
Molecules | Free Full-Text | Halogen–Metal Exchange on Bromoheterocyclics with Substituents Containing an Acidic Proton via Formation of a Magnesium Intermediate | HTML